
The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We will discuss methods for remembering the observed order. For small orbitals (1 s through 3 p), the increase in energy due to n is more significant than the increase due to l however, for larger orbitals the two trends are comparable and cannot be simply predicted. Electrons in orbitals that experience more shielding are less stabilized and thus higher in energy. This phenomenon is called shielding and will be discussed in more detail in the next section. Electrons that are closer to the nucleus slightly repel electrons that are farther out, offsetting the more dominant electron–nucleus attractions slightly (recall that all electrons have −1 charges, but nuclei have + Z charges). Within each shell, as the value of l increases, the electrons are less penetrating (meaning there is less electron density found close to the nucleus), in the order s > p > d > f. But this is not the only effect we have to take into account. Thus, the attraction to the nucleus is weaker and the energy associated with the orbital is higher (less stabilized). As the principal quantum number, n, increases, the size of the orbital increases and the electrons spend more time farther from the nucleus. The filling order is based on observed experimental results, and has been confirmed by theoretical calculations. Thus, many students find it confusing that, for example, the 5 p orbitals fill immediately after the 4 d, and immediately before the 6 s. Such overlaps continue to occur frequently as we move up the chart.įigure 6.24 Generalized energy-level diagram for atomic orbitals in an atom with two or more electrons (not to scale).Įlectrons in successive atoms on the periodic table tend to fill low-energy orbitals first. The 3 d orbital is higher in energy than the 4 s orbital. However, this pattern does not hold for larger atoms. 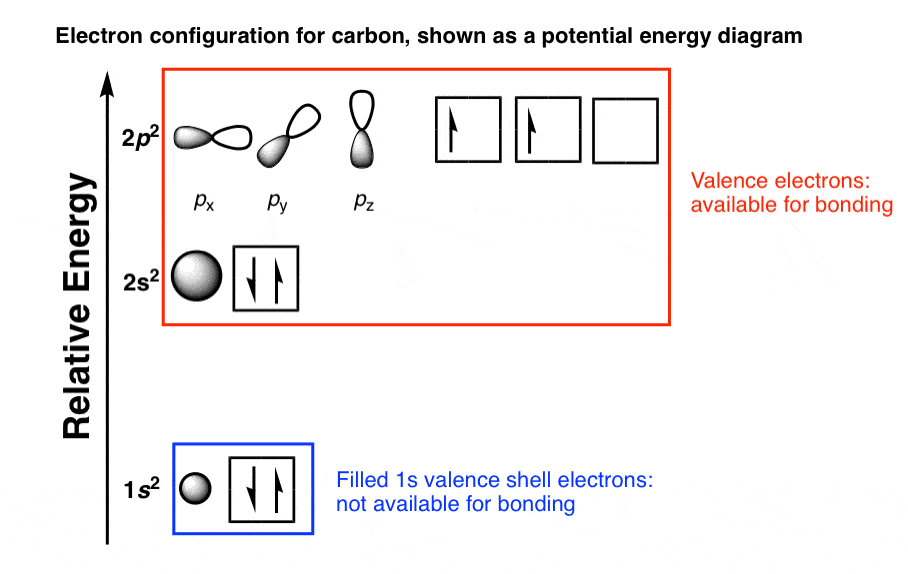
The energy increases as we move up to the 2 s and then 2 p, 3 s, and 3 p orbitals, showing that the increasing n value has more influence on energy than the increasing l value for small atoms. The 1 s orbital at the bottom of the diagram is the orbital with electrons of lowest energy. Figure 6.24 depicts how these two trends in increasing energy relate. In any atom with two or more electrons, the repulsion between the electrons makes energies of subshells with different values of l differ so that the energy of the orbitals increases within a shell in the order s < p < d < f. The energy of atomic orbitals increases as the principal quantum number, n, increases. The specific arrangement of electrons in orbitals of an atom determines many of the chemical properties of that atom. This allows us to determine which orbitals are occupied by electrons in each atom. Having introduced the basics of atomic structure and quantum mechanics, we can use our understanding of quantum numbers to determine how atomic orbitals relate to one another. Relate electron configurations to element classifications in the periodic table.Identify and explain exceptions to predicted electron configurations for atoms and ions.Derive the predicted ground-state electron configurations of atoms.Using the same method, calcium has 2 valence electrons.By the end of this section, you will be able to: For the element aluminum, above, we see 2 electrons in the 3s orbital and 1 electron in the 3p orbital, so aluminum has a total of 3 valence electrons. These electrons are important because they are the ones that are gained, lost or shared in chemical reactions. We typically do not consider “d” electrons as valence electrons and therefore a more specific definition is needed: valence electrons are those electrons in the highest principal energy level. In many cases, “d” electrons will be present after the last noble gas, as in the element manganese : 4s 23d 5. The electrons written after the noble gas in brackets are called valence electrons. These electrons do not participate in chemical reactions. Those electrons in the brackets are called core electrons. 
Valence Electrons-This notation for writing electron configurations helps us to highlight 2 different types of electrons in the atom. 
We may NOT use any element in the brackets, only noble gases.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
